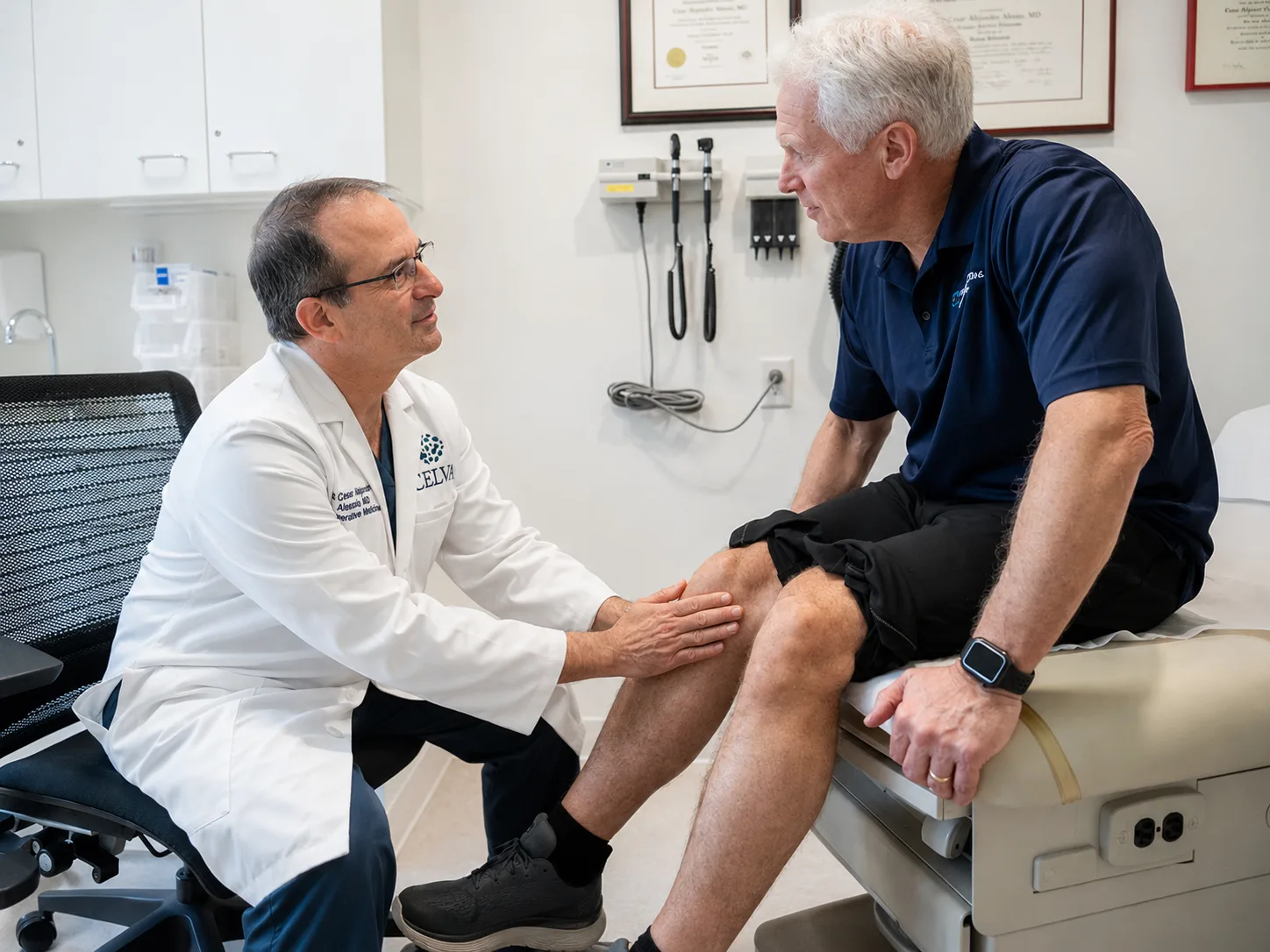
The orthopedist gave me two options. Celva gave me a third one I could live with.
Celva Bio is a physician-led clinic inside Hospital Angeles, Tijuana. We deliver mesenchymal stem cell therapy (the body's own repair-coordinating cells) for joint pain, longevity, and the conditions standard care hasn't solved.

Most patients arrive through one of three tracks. Each is physician-reviewed against the same criteria: indication, imaging, prior treatments tried, and candidacy risk.
Celva operates inside Hospital Angeles, Tijuana, Mexico's most recognized hospital name for 40 years. Twenty minutes from the U.S. border.
Our team handles the day: private transport, border logistics, clinical coordination, aftercare. You handle the recovery.

Every Celva patient moves through the same five-step process. Intake first. The physician team in Mexico assesses candidacy before any treatment is recommended, and a significant share of inquiries are declined.
You fly into San Diego. We take care of the day.
Your first call is with a Celva patient coordinator who gathers your medical history, imaging, prior treatments, and goals. The physician team at Hospital Angeles, Tijuana then reviews your case independently and returns a recommendation.
Concierge pickup from your San Diego hotel. Medical fast-pass crossing. You are accompanied. Never sent alone.
Vitals, informed consent, and a walk-through of the day. Questions welcomed. Intake is not a formality.
MSC infusion (IV) or image-guided targeted injection. Hospital procedure suite, physician supervision throughout.
Concierge transport back to San Diego once the physician team has authorized discharge — most patients are home the same day. Scheduled virtual follow-ups at 30, 60, and 90 days.
Mesenchymal stem cells initiate a signaling cascade that operates at four levels at once. Together these mechanisms address the upstream biology behind chronic conditions — changing the environment so the body's own repair capacity can function again. The signaling persists well beyond the cells themselves.
Allogeneic umbilical-cord MSCs. Screened donors. cGMP-compliant cell processing. Physician supervision at a full hospital. Not a wellness clinic.
Suppresses the chronic inflammatory signaling driving pain, dysfunction, and progressive tissue damage — systemically.
Interrupts the fibrotic cascade that replaces functional tissue with scar, a downstream consequence of chronic inflammation.
Recalibrates dysregulated immune activity across the body without systemic immune suppression.
Supports formation of new vascular networks, improving tissue perfusion and creating a more favorable systemic repair environment.
Representative cases from our clinical follow-up records. De-identified. Verified with patient consent. Timeline is the patient's own.

The orthopedist gave me two options. Celva gave me a third one I could live with.

I stopped waiting for something to go wrong before doing anything about it.
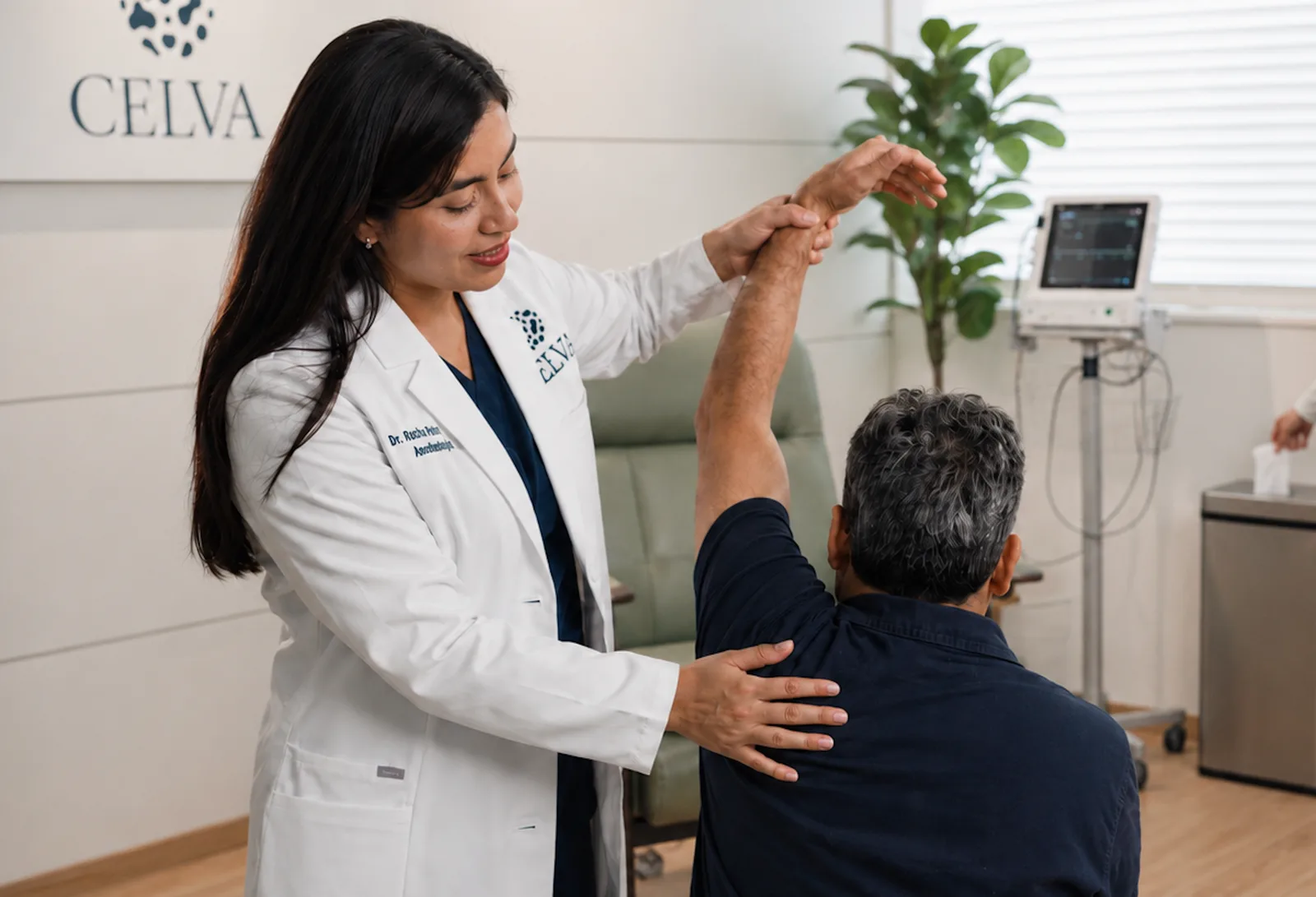
The surgeon said shave-and-hope. I wanted a different conversation.
Most patients research for weeks before reaching out. These are the questions we hear on almost every first call. Straight answers. Not reassurances.
No pressure. No commitments. Your first call is with a Celva patient coordinator who collects your information and passes it to the physician team in Mexico for independent review. Bring every question you have — about your condition, the treatment, the process, and whether Celva is the right fit.